
Chemistry, 30.07.2019 21:10, firenation18
A50.0 ml sample of an aqueous h2so4 solution is titrated with a 0.375 m naoh solution. the equivalence point is reached with 62.5 ml of the base. the concentration of h2so4 is m.

Answers: 3
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 19:00, georgesarkes12
Mercury metal is poured into a graduated cylinder that holds exactly 22.5 ml the mercury used to fill the cylinder mass in 306.0 g from this information calculate the density of mercury
Answers: 2
Do you know the correct answer?
A50.0 ml sample of an aqueous h2so4 solution is titrated with a 0.375 m naoh solution. the equivalen...
Questions in other subjects:

Mathematics, 17.01.2020 04:31

Mathematics, 17.01.2020 04:31





Computers and Technology, 17.01.2020 04:31




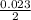 = 0.012mole of acid
= 0.012mole of acid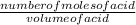
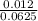 = 0.188M
= 0.188M 



