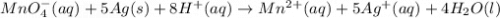Chemistry, 30.07.2019 03:20, andrew5182
Balance the redox reaction equation (occurring in acidic solution) and choose the correct coefficients for each reactant and product. __mno4−(aq)+ag(s)+h+(aq)→mn2+(aq)+a g+(aq)+__h2o(l)

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:00, sleimanabir
Balance this equation: n2 + h2 > nh3, write the following molar ratios: n2 / n2 / nh3 h2 /
Answers: 1

Chemistry, 22.06.2019 19:20, halledoll2002
Anyone who's in connections academy chemistry b have the factors that affect the rate of a reaction portfolio already done?
Answers: 3


Chemistry, 22.06.2019 21:30, leenzazou587
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1
Do you know the correct answer?
Balance the redox reaction equation (occurring in acidic solution) and choose the correct coefficien...
Questions in other subjects:


Mathematics, 17.05.2021 19:30


Advanced Placement (AP), 17.05.2021 19:30

History, 17.05.2021 19:30


Mathematics, 17.05.2021 19:30

English, 17.05.2021 19:30

Mathematics, 17.05.2021 19:30

Mathematics, 17.05.2021 19:30


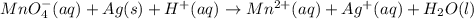
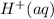 on reactant side by 8 and multiply
on reactant side by 8 and multiply 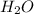 on product side by 4.
on product side by 4.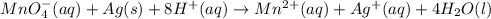
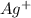 on product side by 5.
on product side by 5.