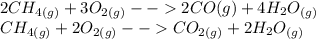Chemistry, 30.07.2019 03:10, wwwserban9281
Write the two balanced equations, one for the incomplete combustion of methane (to form carbon monoxide and water vapor), and another one for the complete combustion of methane (forming carbon dioxide and water vapor). what are the conditions, at which do you think the incomplete combustion might take place instead of the complete combustion? what is the health risk associated with this process?

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 23:50, josie311251
Be sure to answer all parts. the following equilibrium constants were determined at 1123 k: c(s) + co2(g) ⇌ 2co(g) k'p = 1.30 × 1014 co(g) + cl2(g) ⇌ cocl2(g) k''p = 6.00 × 10−3 calculate the equilibrium constant at 1123 k for the reaction: c(s) + co2(g) + 2cl2(g) ⇌ 2cocl2(g) 4.68 × 10 9 (enter your answer in scientific notation.) write the equilibrium constant expression, kp:
Answers: 3

Chemistry, 23.06.2019 16:20, chimwim8347
Select the correct answer. what is the wavelength of radio waves? a. between 1 x 10-3 and 1 x 10-1 meters b. less than 1 x 10-11 meters c. more than 1 x 10-1 meters d. between 1 x 10-8 - 4 x 10-7 meters e. between 4 x 10-7 - 7 x 10-7 meters
Answers: 3
Do you know the correct answer?
Write the two balanced equations, one for the incomplete combustion of methane (to form carbon monox...
Questions in other subjects:

History, 16.11.2020 20:20


Mathematics, 16.11.2020 20:20

Mathematics, 16.11.2020 20:20


Biology, 16.11.2020 20:20

Physics, 16.11.2020 20:20



Mathematics, 16.11.2020 20:20