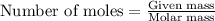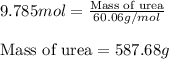Chemistry, 25.07.2019 01:20, 1233042260
The emission of no2 by fossil fuel combustion can be prevented by injecting gaseous urea into the combustion mixture. the urea reduces no (which oxidizes in air to form no2) according to the following reaction: 2co(nh2)2(g)+4no(g)+o2(g)→4n2(g)+2c o2(g)+4h2o(g) suppose that the exhaust stream of an automobile has a flow rate of 2.32 l/s at 670 k and contains a partial pressure of no of 12.1 torr. what total mass (in gram) of urea is necessary to react completely with the no formed during 8.1 hours of driving?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 23.06.2019 04:31, 24swimdylanoh
What are the coefficients that will balance the skeleton equation below? n2 + h2 → nh3
Answers: 1



Do you know the correct answer?
The emission of no2 by fossil fuel combustion can be prevented by injecting gaseous urea into the co...
Questions in other subjects:



Mathematics, 04.03.2021 19:00

Mathematics, 04.03.2021 19:00

Mathematics, 04.03.2021 19:00



History, 04.03.2021 19:00


Mathematics, 04.03.2021 19:00


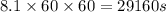
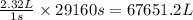
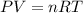
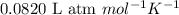
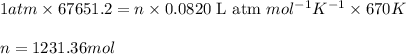
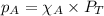
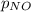 = partial pressure of NO = 12.1 torr
= partial pressure of NO = 12.1 torr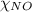 = mole fraction of NO = ?
= mole fraction of NO = ?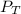 = total pressure of solution = 1 atm = 760 torr (Conversion factor: 1 atm = 760 torr)
= total pressure of solution = 1 atm = 760 torr (Conversion factor: 1 atm = 760 torr)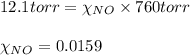
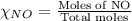
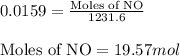
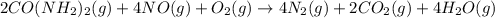
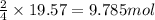 of urea.
of urea.