Chemistry, 24.07.2019 22:30, adejumoayobami1
The radioactive isotope of lead, pb-209, decays at a rate proportional to the amount present at time t and has a half-life of 3.3 hours. if 1 gram of this isotope is present initially, how long will it take for 75% of the lead to decay? (round your answer to two decimal places.)

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 14:20, kingaman
Which of the following statements is not true? • a. covalent compounds have low melting and boiling points. • ob. covalent bonds between atoms of a compound are relatively weak compared to bonds between molecules. • c. covalent bonds occur between nonmetals. • d. covalent compounds are often gases or liquids.
Answers: 2



Chemistry, 23.06.2019 07:30, isalih7256
In the diagram here that represents the reaction, which reactant, a or b, is the limiting reagent?
Answers: 1
Do you know the correct answer?
The radioactive isotope of lead, pb-209, decays at a rate proportional to the amount present at time...
Questions in other subjects:

Mathematics, 03.01.2021 17:50

Law, 03.01.2021 17:50


Social Studies, 03.01.2021 17:50

Mathematics, 03.01.2021 17:50




Mathematics, 03.01.2021 17:50


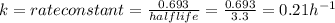
![time=\frac{1}{k}(ln[\frac{A_{0}}{A_{t}}]](/tpl/images/0128/8405/a5dc9.png)