
Chemistry, 21.07.2019 07:10, Kelshonti15
Asample of potassium phosphate octahydrate (k3po4•8h2o) is heated until 7.93 grams of water are released. how many grams did the original hydrate weigh?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:10, andybiersack154
Explain the relationship between forward and backward reactions in equilibrium, and predict how changing the amount of a reactant (creating a tension) will affect that relationship.
Answers: 1


Chemistry, 22.06.2019 11:40, Wemaybewrong
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1

Chemistry, 22.06.2019 13:00, netflixacc0107
Amixture with the same composition throughout is!
Answers: 1
Do you know the correct answer?
Asample of potassium phosphate octahydrate (k3po4•8h2o) is heated until 7.93 grams of water are rele...
Questions in other subjects:



History, 12.12.2020 16:40



Mathematics, 12.12.2020 16:40


Mathematics, 12.12.2020 16:40



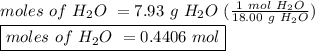

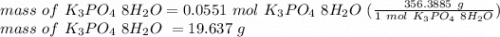
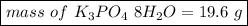
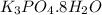
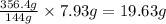 of salt is heated.
of salt is heated.



