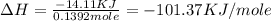Chemistry, 06.07.2019 19:10, angelZ3947
Find the enthalpy of neutralization of hcl and naoh. 87 cm3 of 1.6 mol dm-3 hydrochloric acid was neutralized by 87 cm3 of 1.6 mol dm-3 naoh. the temperature rose from 298 k to 317.4 k. the specific heat capacity is the same as water, 4.18 j/k g.
a. -101.37 kj
b. -7.05 kj
c. 7055 kj
d. 10,1365 kj

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 16:10, 00015746
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2


Chemistry, 23.06.2019 02:00, raulflores01
Which best describes the present-day universe? opaque, expanding very slowly, stars produce heavy elements transparent, expanding at an accelerated rate, stars produce heavy elements opaque, expanding at an accelerated rate, stars produce only hydrogen and helium transparent, expanding very slowly, stars produce only hydrogen and helium
Answers: 1
Do you know the correct answer?
Find the enthalpy of neutralization of hcl and naoh. 87 cm3 of 1.6 mol dm-3 hydrochloric acid was ne...
Questions in other subjects:




Mathematics, 07.01.2021 04:10


Mathematics, 07.01.2021 04:10

Mathematics, 07.01.2021 04:10

Mathematics, 07.01.2021 04:10



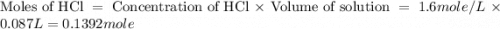
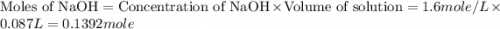

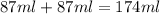
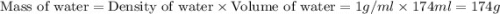
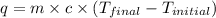
 = specific heat of water =
= specific heat of water = 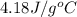
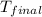 = final temperature of water = 317.4 K
= final temperature of water = 317.4 K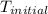 = initial temperature of metal = 298 K
= initial temperature of metal = 298 K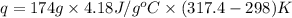
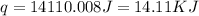
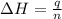
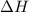 = enthalpy of neutralization = ?
= enthalpy of neutralization = ?