
For each of the following acid-base reactions, calculate how many grams of each acid are necessary to completely react with and neutralize 2.7 g of the base. part a : hcl(aq)+naoh(aq)→h2o(l)+nacl(aq)par t b : 2hno3(aq)+ca(oh)2(aq)→2h2o(l)+ca(no 3)2(aq)part c : h2so4(aq)+2koh(aq)→2h2o(l)+k2so4(aq )

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:00, ayoismeisalex
Matches the chemical name of each oxide of phosphorus to its chemical formula
Answers: 2

Chemistry, 22.06.2019 12:30, masteroftheuniverse3
When a scientific theory has been tested and proved by the scientific community, it becomes a law
Answers: 2

Chemistry, 22.06.2019 13:30, xojade
Which statements are true concerning mineral formation? check all that apply. the slower the cooling, the larger the crystals. the faster the cooling, the smaller the crystals. crystals formed from magma are smaller than crystals formed from lava. minerals can only form in solutions when the solution is heated deep underground. when a solution cools, elements and compounds leave the solution and crystallize as minerals. minerals formed from hot water solutions can form narrow channels in the surrounding rock.
Answers: 1
Do you know the correct answer?
For each of the following acid-base reactions, calculate how many grams of each acid are necessary t...
Questions in other subjects:

Mathematics, 16.09.2020 14:01

Mathematics, 16.09.2020 14:01

Mathematics, 16.09.2020 14:01

Mathematics, 16.09.2020 14:01

Mathematics, 16.09.2020 14:01

Mathematics, 16.09.2020 14:01

Mathematics, 16.09.2020 14:01

Mathematics, 16.09.2020 14:01

Mathematics, 16.09.2020 14:01

Mathematics, 16.09.2020 14:01


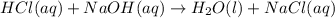
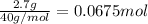
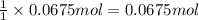 of HCl.
of HCl.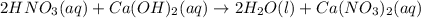
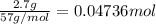
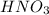 neutralizes with 1 mol of
neutralizes with 1 mol of 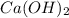 .
.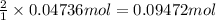 of
of 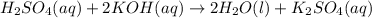
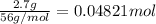
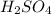 neutralizes with 2 mol of
neutralizes with 2 mol of 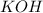 .
.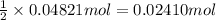 of
of 



