
Chemistry, 05.07.2019 05:20, Mw3spartan17
Which of the following values would you expect for the ratio of half-lives for a reaction with starting concentrations of 0.05m and 0.01m, t1/2(0.05m) / t1/2 (0.01m), if a reaction is known to be zero order?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:00, marissastewart533
City a and city b had two different temperatures on a particular day. on that day, four times the temperature of city a was 8â° c more than 3 times the temperature of city b. the temperature of city a minus twice the temperature of city b was â’3â° c. what was the temperature of city a and city b on that day? city a was 5â° c, and city b was 4â° c. city a was 3â° c, and city b was â’1â° c. city a was 8â° c, and city b was â’3â° c. city a was 5â° c, and city b was â’5â° c.
Answers: 2

Chemistry, 22.06.2019 01:40, georgehall3027
C3h8o3 - glycerol major species present when dissolved in water
Answers: 2


Chemistry, 22.06.2019 16:00, julesperez22
Click the button that shows the correct relationship of the electron affinities of the elements sodium and phosphorus. sodium’s electron affinity value is more negative than the electron affinity value of phosphorus. phosphorus’ electron affinity value is more negative than the electron affinity value of sodium. this information cannot be determined using the periodic table. answer is b on e2020.
Answers: 3
Do you know the correct answer?
Which of the following values would you expect for the ratio of half-lives for a reaction with start...
Questions in other subjects:

Mathematics, 04.04.2020 01:51




Arts, 04.04.2020 01:51



Mathematics, 04.04.2020 01:51



![k=\frac{1}{t}([A_o]-[A])](/tpl/images/0052/8982/5bd85.png)
![[A_o]](/tpl/images/0052/8982/dc622.png) = initial concentration
= initial concentration![[A]=\frac{1}{2}[A_o]](/tpl/images/0052/8982/13a37.png)
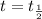 , the equation (1) becomes:
, the equation (1) becomes:![t_{\frac{1}{2}}=\frac{[A_o]}{2k}](/tpl/images/0052/8982/9ef84.png)
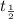
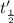
![\frac{t_{\frac{1}{2}}}{t_{\frac{1}{2}}'}=\frac{\frac{[0.05 M]}{2k}}{\frac{[0.01 M]}{2k}}=\frac{5}{1}](/tpl/images/0052/8982/a48c0.png)




