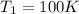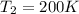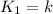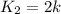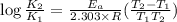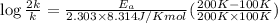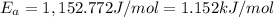Chemistry, 03.07.2019 23:40, afropenguin7371
We know that for a given reaction when the temperature increases from 100 k to 200 k the rate constant doubles. what is the activation energy in kj/mol

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 00:10, mpchop
According to the diagram; a) identify the anode of the cell and write the half-reaction that occurs there b) write the overall equation for the reaction that occurs as the cell operates c) calculate the value of the standard cell potential ,e cell. d)write the shorthand notation of the cell above e)indicate the flow of the electrons on the diagram
Answers: 3

Chemistry, 22.06.2019 14:50, chem1014
Given the following information: mass of proton = 1.00728 amu mass of neutron = 1.00866 amu mass of electron = 5.486 × 10^-4 amu speed of light = 2.9979 × 10^8 m/s calculate the nuclear binding energy (absolute value) of 3li^6. which has an atomic mass of 6.015126 amu. j/mol.
Answers: 2

Chemistry, 22.06.2019 18:30, ashleymer384
Two people each hold the end of a rope and create waves by moving their arms up and down. this wave is best classified as a transverse wave because a) both the rope particles and the wave are moving in the same direction. b) the wave is moving up and down as the particles of the rope move horizontally. c) the wave is moving horizontally as the particles of the rope move up and down. eliminate d) the wave is moving in a parallel direction with the motion of the person's arms.
Answers: 3
Do you know the correct answer?
We know that for a given reaction when the temperature increases from 100 k to 200 k the rate consta...
Questions in other subjects:


Mathematics, 29.10.2021 04:10

Mathematics, 29.10.2021 04:10

Mathematics, 29.10.2021 04:10



Social Studies, 29.10.2021 04:10

Biology, 29.10.2021 04:10

History, 29.10.2021 04:10

History, 29.10.2021 04:10