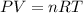Nitrogen gas is introduced into a large deflated plastic bag. no gas is allowed to escape, but as more and more nitrogen is added, the bag inflates to accommodate it. the pressure of the gas within the bag remains at 1.00 atm and its temperature remains at room temperature (20.0∘c). how many moles n have been introduced into the bag by the time its volume reaches 22.4 l

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, alexisgoss8091
Which traits do human embryos have that link them to a common ancestor with fish and reptiles? a. scales and tail b. gill slits and scales c. tail and gill slits d. hair and tail
Answers: 2



Chemistry, 22.06.2019 22:20, trockout4868
How do cfcs cause ozone depletion? how do cfcs cause ozone depletion? ultraviolet radiation breaks down cfcs, molecules containing chlorine. chlorine then breaks one oxygen atom away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation breaks down cfcs, molecules containing chlorine. chlorine then breaks two oxygen atoms away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation creates cfcs, molecules containing chlorine. chlorine then breaks two oxygen atoms away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation creates cfcs, molecules containing chlorine. chlorine then breaks one oxygen atom away from ozone, leaving behind a paired oxygen molecule.
Answers: 2
Do you know the correct answer?
Nitrogen gas is introduced into a large deflated plastic bag. no gas is allowed to escape, but as mo...
Questions in other subjects:



Mathematics, 09.12.2019 03:31

Mathematics, 09.12.2019 03:31




Arts, 09.12.2019 03:31