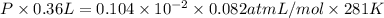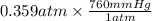Chemistry, 26.06.2019 04:20, silviamgarcia
The vapor pressure of liquid acetone, ch3coch3, is 100 mm hg at 281 k. a 6.06e-2 g sample of liquid ch3coch3 is placed in a closed, evacuated 360. ml container at a temperature of 281 k. calculate what the ideal gas pressure would be in the container if all of the liquid acetone evaporated.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:50, giiffnlojd
Using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 pb+2(aq) + 2cl -(aq). if the value of ksp was determined to be only 1.2 x 10-2: too much solid has dissolved. additional precipitate is forming. the solution is unsaturated. the ions are now combining to reduce their concentrations.
Answers: 3


Do you know the correct answer?
The vapor pressure of liquid acetone, ch3coch3, is 100 mm hg at 281 k. a 6.06e-2 g sample of liquid...
Questions in other subjects:

Mathematics, 02.04.2020 08:58

Advanced Placement (AP), 02.04.2020 08:58

Mathematics, 02.04.2020 08:58

Mathematics, 02.04.2020 08:58

English, 02.04.2020 08:59


Chemistry, 02.04.2020 08:59

Mathematics, 02.04.2020 08:59

Mathematics, 02.04.2020 08:59

Geography, 02.04.2020 08:59


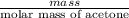
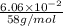
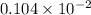 mole
mole