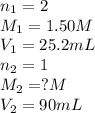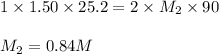Chemistry, 28.01.2020 11:31, covergurllaa
Avolume of 90.0 ml of aqueous potassium hydroxide (koh) was titrated against a standard solution of sulfuric acid (h2so4). what was the molarity of the koh solution if 25.2 ml of 1.50 m h2so4 was needed? the equation is 2koh(aq)+h2so4(aq)→k2so4(aq)+2h2o(l )

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:00, pandasarecute53
If the density of water is 1.0 g/cm3, which of these materials would float in water, based on their densities? check all that apply. aluminum cork iron lead wax
Answers: 1

Chemistry, 22.06.2019 11:30, ayoismeisjjjjuan
Which statement best describes the flow of energy in this scenario
Answers: 1
Do you know the correct answer?
Avolume of 90.0 ml of aqueous potassium hydroxide (koh) was titrated against a standard solution of...
Questions in other subjects:

Mathematics, 26.02.2021 19:20

Mathematics, 26.02.2021 19:20

Spanish, 26.02.2021 19:20



Geography, 26.02.2021 19:20


Computers and Technology, 26.02.2021 19:20



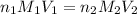
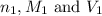 are the n-factor, molarity and volume of acid which is
are the n-factor, molarity and volume of acid which is 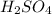
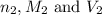 are the n-factor, molarity and volume of base which is KOH.
are the n-factor, molarity and volume of base which is KOH.