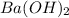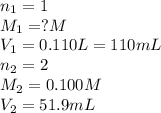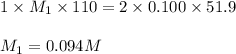Chemistry, 10.01.2020 11:31, mpzpowell7506
Consider the neutralization reaction 2hno3(aq)+ba(oh)2(aq)⟶2h2o(l)+ba(no 3)2(aq) 2hno3(aq)+ba(oh)2(aq)⟶2h2o(l)+ba(no 3)2(aq) a 0.110 l0.110 l sample of an unknown hno3hno3 solution required 51.9 ml51.9 ml of 0.100 m ba(oh)20.100 m ba(oh)2 for complete neutralization. what is the concentration of the hno3hno3 solution? concentration:

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 23.06.2019 14:00, GreenHerbz206
During an acid-base titration, when do the contents of the beaker consist of only water, a salt, and a trace of indicator?
Answers: 2
Do you know the correct answer?
Consider the neutralization reaction 2hno3(aq)+ba(oh)2(aq)⟶2h2o(l)+ba(no 3)2(aq) 2hno3(aq)+ba(oh)2(a...
Questions in other subjects:


Mathematics, 05.05.2021 21:00

Arts, 05.05.2021 21:00



Chemistry, 05.05.2021 21:00

History, 05.05.2021 21:00

History, 05.05.2021 21:00

Mathematics, 05.05.2021 21:00

Chemistry, 05.05.2021 21:00


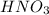 solution will be 0.094 M.
solution will be 0.094 M.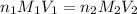
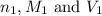 are the n-factor, molarity and volume of acid which is
are the n-factor, molarity and volume of acid which is 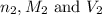 are the n-factor, molarity and volume of base which is
are the n-factor, molarity and volume of base which is