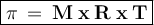Chemistry, 04.02.2020 12:51, mathsux6600
Adextrose/saline aqueaout solution doctors commonly use to replace fluids in the body contains 1.75g/l nacl and 40.0g/l dextrose. a) what is the molarity of all solutes in this solution? b) what is the osmotic pressure of the solution at 25 c. assume total dissociation of nacl. show work !

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:30, nekathadon
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1


Chemistry, 22.06.2019 16:30, montanolumpuy
How many moles of sulfuric acid (h2so4) are needed to react completely with 6.8 moles of lithium hydroxide (lioh)? 2lioh + h2so4 → li2so4 + 2h2o a. 3.4 mol h2so4b. 6.8 mol h2so4 c. 10.2 mol h2so4 d. 13.6 mol h2so4
Answers: 3

Chemistry, 22.06.2019 22:10, preachersgirl5
What is the indicator of the number of ions in solution? the amount of conductivity the amount of precipitate the amount of solute added
Answers: 1
Do you know the correct answer?
Adextrose/saline aqueaout solution doctors commonly use to replace fluids in the body contains 1.75g...
Questions in other subjects:







Mathematics, 13.04.2020 16:40

Mathematics, 13.04.2020 16:40


English, 13.04.2020 16:40