
The explosive nitroglycerin (c3h5n3o9) decomposes rapidly upon ignition or sudden impact according to the following balanced equation: 4 c3h5n3o9 (l) → 12 co2 (g) + 10 h2o (g) + 6 n2 (g) + o2 (g) δrxnho = −5678 kj calculate the standard enthalpy of formation (δfho) for nitroglycerin. the enthalpy of formation of co2 (g) is -393.5 kj/mol. the enthalpy of formation of h2o (g) is -241.8 kj/mol.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:00, shradhwaip2426
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3



Chemistry, 22.06.2019 21:50, donttrip10
What is a main difference between a mixture and a pure substance? a mixture is only a liquid, but a pure substance can be in any state. a mixture looks the same throughout, but a pure substance does not.1 a mixture can vary in composition, but a pure substance has a set composlo a mixture can be made up of a single compound, but a pure substance car
Answers: 2
Do you know the correct answer?
The explosive nitroglycerin (c3h5n3o9) decomposes rapidly upon ignition or sudden impact according t...
Questions in other subjects:

English, 13.09.2020 06:01

Chemistry, 13.09.2020 06:01

Mathematics, 13.09.2020 06:01

Mathematics, 13.09.2020 06:01

Mathematics, 13.09.2020 06:01

Mathematics, 13.09.2020 06:01

Mathematics, 13.09.2020 06:01

Mathematics, 13.09.2020 06:01

Mathematics, 13.09.2020 06:01

Mathematics, 13.09.2020 06:01


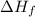 for
for 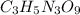 in the reaction is -365.5 kJ/mol.
in the reaction is -365.5 kJ/mol.![\Delta H_{rxn}=\sum [n\times \Delta H_f_{(product)}]-\sum [n\times \Delta H_f_{(reactant)}]](/tpl/images/0445/1737/18a63.png)
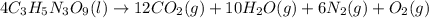
![\Delta H_{rxn}=[(12\times \Delta H_f_{(CO_2)})+(10\times \Delta H_f_{(H_2O)})+(6\times \Delta H_f_{(N_2)})+(1\times \Delta H_f_{(O_2)})]-[(4\times \Delta H_f_{(C_3H_5N_3O_9)})]](/tpl/images/0445/1737/c0f93.png)
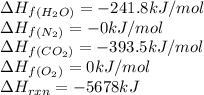
![-5678=[(12\times (-393.5))+(10\times (-241.8))+(6\times (0))+(1\times (0))]-[(4\times \Delta H_f_{(C_3H_5N_3O_9)})]\\\\\Delta H_f_{(C_3H_5N_3O_9)}=-365.5kJ/mol](/tpl/images/0445/1737/dcb72.png)




