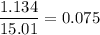Raoult's law states that for a given solution, the partial pressure of each component is equal to the mole fraction in that solution. The vapor pressure of the solution is 22 mmHg.
The vapor pressure is defined as the force exerted by the vapors in the walls of a container. It is a colligative property, such that the amount of substance increased or decreased is dependent on the amount of solute present.
The equation can be represented as:
P
- P
=
P = X

x P
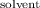
where,
P
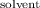
= vapor pressure of the pure solventP
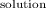
= vapor pressure of the solutionX

= molar fraction of the solute
Now, calculating the mole fraction, where:
Moles of ions = 2 × number of moles of KCl = 1.134 molNumber of moles KCl =
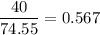
molesTotal moles are: 1.134 mol + 13.877 mol = 15.011 molX

=
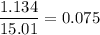
Now, the vapor pressure of the solution can be calculated as:
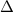 P = X
P = X x P
x P
 P = 0.0755 × 23.76 mmHg = 1.78 mmHg
P = 0.0755 × 23.76 mmHg = 1.78 mmHg
Hence, the vapor pressure of the solution:
P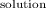 =
= 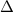 P + P
P + P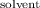
P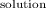 = 23.76 mmHg - 1.79 mm Hg = 21.97mmHg
= 23.76 mmHg - 1.79 mm Hg = 21.97mmHg
Therefore, the vapor pressure of the solution is approximately 22 mmHg.
To know more about vapor pressure, refer to the following link:
link
















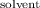 - P
- P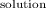 =
= 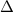 P = X
P = X  x P
x P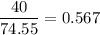 molesTotal moles are: 1.134 mol + 13.877 mol = 15.011 molX
molesTotal moles are: 1.134 mol + 13.877 mol = 15.011 molX