
Chemistry, 25.06.2019 12:10, coltonwsmith
During a laboratory experiment, a 2.36-gram sample of nahco3 was thermally decomposed. in this experiment, carbon dioxide and water vapors escape and are combined to form carbonic acid. after decomposition, the sample weighed 1.57 grams. calculate the percentage yield of carbonic acid for the reaction. describe the calculation process in detail.

Answers: 1
Other questions on the subject: Chemistry




Chemistry, 22.06.2019 23:30, ninilizovtskt
If maltose undergoes hydrolysis what subunits does it results to?
Answers: 2
Do you know the correct answer?
During a laboratory experiment, a 2.36-gram sample of nahco3 was thermally decomposed. in this exper...
Questions in other subjects:


Mathematics, 17.09.2019 14:30

Physics, 17.09.2019 14:30



Social Studies, 17.09.2019 14:30

History, 17.09.2019 14:30

Mathematics, 17.09.2019 14:30

History, 17.09.2019 14:30

Biology, 17.09.2019 14:30


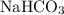 decomposes to produce one
decomposes to produce one 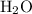 molecule and one
molecule and one 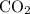 molecule.
molecule.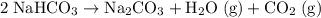 .
.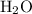 molecule combines with one
molecule combines with one 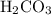 molecule.
molecule.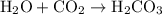 .
.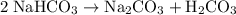 .
. .
.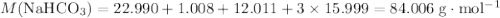 .
.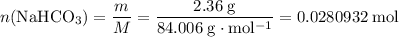 .
.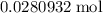 of
of 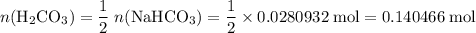 .
.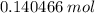 of
of 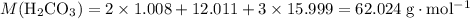 .
.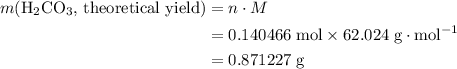 .
.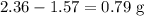 in this experiment. However, mass is conserved in chemical reactions. The missing
in this experiment. However, mass is conserved in chemical reactions. The missing 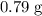 must be the mass of the gaseous
must be the mass of the gaseous 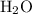 and
and 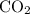 that have escaped from the container.
that have escaped from the container.  .
.



