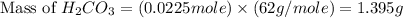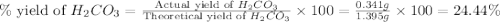Chemistry, 25.06.2019 17:10, sweetbri7p5v6tn
During a laboratory experiment, a 3.81-gram sample of nahco3 was thermally decomposed. in this experiment, carbon dioxide and water vapors escape and are combined to form carbonic acid. after decomposition, the sample weighed 2.86 grams. calculate the percentage yield of carbonic acid for the reaction. describe the calculation process in detail. nahco3 → na2co3 + h2co3

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:00, krystalhurst97
How are isotopes of the same chemical element alike? how are they different?
Answers: 1



Chemistry, 22.06.2019 20:30, Schoolworkspace453
Consider the following unbalanced equation for the combustion of hexane: αc6h14(g)+βo2(g)→γco2(g)+δh2o(g) part a balance the equation. give your answer as an ordered set of numbers α, β, γ, use the least possible integers for the coefficients. α α , β, γ, δ = nothing request answer part b determine how many moles of o2 are required to react completely with 5.6 moles c6h14. express your answer using two significant figures. n n = nothing mol request answer provide feedback
Answers: 2
Do you know the correct answer?
During a laboratory experiment, a 3.81-gram sample of nahco3 was thermally decomposed. in this exper...
Questions in other subjects:







Mathematics, 27.10.2020 18:00



Chemistry, 27.10.2020 18:00


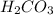 is, 24.44 %
is, 24.44 %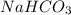 .
.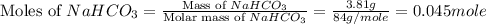
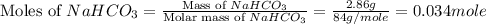
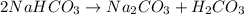
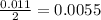 mole of
mole of 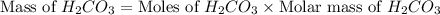
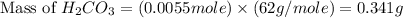
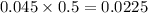 mole of
mole of