
Chemistry, 29.06.2019 08:20, davelopez979
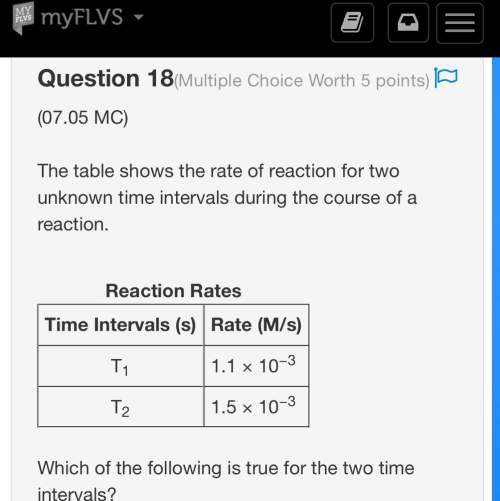
*worth 30 points + brainliest * the table shows the rate of reaction for two unknown time intervals during the course of a reaction. reaction rates time intervals (s) rate (m/s) t1 1.1 × 10−3 t2 1.5 × 10−3 which of the following is true for the two time intervals? a. t1 is longer than t2, and the concentration of reactants at the end of t1 is lower than that of t2. b. t1 is longer than t2, and the concentration of products the end of t1 is lower than that of t2. c. t1 is shorter than t2, and the concentration of reactants at the end of t1 is higher than that of t2. d. t1 is shorter than t2, and the concentration of products at the end of t1 is higher than that of t2.

Answers: 1
Similar questions

Chemistry, 25.06.2019 08:30, ngoziblack
Answers: 2

Chemistry, 28.06.2019 11:20, michaeladkins1979
Answers: 2

Chemistry, 12.08.2019 23:20, sand1g67
Answers: 1

Chemistry, 26.09.2019 06:10, samtuck33
Answers: 2
Do you know the correct answer?
*worth 30 points + brainliest * the table shows the rate of reaction for two unknown time intervals...
Questions in other subjects:



History, 11.07.2021 22:30

Engineering, 11.07.2021 22:30





Social Studies, 11.07.2021 22:30






