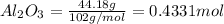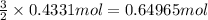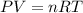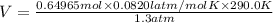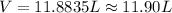Chemistry, 29.06.2019 10:20, gonzalesrosalinda66
During a laboratory experiment, 44.18 grams of al2o3 was formed when o2 reacted with aluminum metal at 290.0 k and 1.3 atm. what was the volume of o2 used during the experiment? 3o2 + 4al → 2al2o3 a. 10.10 liters b. 11.90 liters c. 12.51 liters d. 15.55 liters

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:00, JJlover1892
Ascientist measures the speed of sound in a monatomic gas to be 449 m/s at 20∘c. what is the molar mass of this gas?
Answers: 2

Chemistry, 22.06.2019 14:50, jonmorton159
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3

Chemistry, 22.06.2019 22:30, SavageKidKobe
Rank the four gases (air, exhaled air, gas produced from from decomposition of h2o2, gas from decomposition of nahco3) in order of increasing concentration of co2
Answers: 1

Chemistry, 23.06.2019 02:00, Turtlelover05
Pinene is an unsaturated hydrocarbon found in pine resin. if pinene has m+ = 136 and contains 1 double bond(s) and 2 ring(s); what is its molecular formula? enter the formula in the form ch first, then all other atoms in alphabetical order; do not use subscripts. the formula is case-sensitive
Answers: 3
Do you know the correct answer?
During a laboratory experiment, 44.18 grams of al2o3 was formed when o2 reacted with aluminum metal...
Questions in other subjects:




Chemistry, 24.04.2020 18:35



English, 24.04.2020 18:35

Physics, 24.04.2020 18:35

Mathematics, 24.04.2020 18:35

History, 24.04.2020 18:35


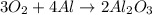
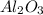 = 44.18 g
= 44.18 g