
Chemistry, 02.02.2020 12:49, paigefields2578
Formic acid, hcooh, is a weak acid present in the venom of red-ants. at equilibrium, [hcooh] = 2.00 m, [hcoo− ] = 4.0 × 10− 1 m, and [h3o+ ] = 9.0 × 10− 4 m.
a. write the equilibrium expression for the ionization

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:30, BornAdopted21
Which piece of equipment would me most useful for measuring the volume of some water? a. pan balance b. graduated cylinder c. tweezers d. flask quick
Answers: 2

Chemistry, 22.06.2019 06:00, rebeccacruzz2017
Calculate - analysis of compound composed of iron and oxygen yields 174.86 of fe and 75.14g of o. what is the empirical formula for this compound?
Answers: 3

Chemistry, 22.06.2019 14:00, coylenoah0
How many absorptions would you expect to observe in the 13c nmr spectra of the following molecules? a) 3-chloropentane b) cis-4-methyl-2-pentene
Answers: 2

Chemistry, 22.06.2019 19:00, Farhan54019
Which change to the system wood cause the freely-moving piston to lower?
Answers: 1
Do you know the correct answer?
Formic acid, hcooh, is a weak acid present in the venom of red-ants. at equilibrium, [hcooh] = 2.00...
Questions in other subjects:

History, 29.06.2019 20:00



Chemistry, 29.06.2019 20:00

Mathematics, 29.06.2019 20:00


History, 29.06.2019 20:00



English, 29.06.2019 20:00


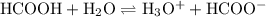
![K_{eq} = \dfrac{[\text{Products}]}{[\text{Reactants}]}](/tpl/images/0492/9894/57030.png)
![K_{eq} = \dfrac{[\text{HCOO}^{-}][\text{H}_{3}\text{O}^{+}]}{[\text{HCOOH}]}](/tpl/images/0492/9894/43500.png)




