
Chemistry, 01.02.2020 07:45, sbhunsaker9025
How many atoms of n are in 137.0 grams of n2o3?
a.
1.085 × 1023
b.
1.802 × 1023
c.
5.985 × 1023
d.
2.171 × 1024
e.
3.604 × 1024

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:00, darg3990rgp2t0r2
Does anyone know a lot about how to: - calculate mass of magnesium metal - calculate the actual yield of magnesium oxide - calculate the theoretical yield of mgo - calculate the percent yield of mgo - determine the percent yield of mgo - determine the average percent yield of mgo i had to do an online lab and its asking these questions but i have no idea where to start or how to be able to find these things. i can post the chart of the data from the lab or if you can tell me exactly how i can find each.
Answers: 3

Chemistry, 22.06.2019 16:50, struckedblazing
Answer asap need it by wednesday morning calculate the ph of 0.02m hcl best answer will be brainliest
Answers: 1

Chemistry, 23.06.2019 04:20, milkshakegrande101
The equation below shows the reaction of zinc with hydrochloric acid (hcl). zn (s) + 2 hcl (aq) —> zncl2 (aq) + h2 (g) what will happen if the concentration of hcl is decreased? a. more zncl2 will be produced. b. the reaction rate will slow down. c. the hydrochloric acid will become more acidic. d. the reaction will produce water instead of hydrogen gas.
Answers: 1
Do you know the correct answer?
How many atoms of n are in 137.0 grams of n2o3?
a.
1.085 × 1023
b...
a.
1.085 × 1023
b...
Questions in other subjects:



Social Studies, 19.03.2021 20:30


Mathematics, 19.03.2021 20:30

Health, 19.03.2021 20:30

Mathematics, 19.03.2021 20:30




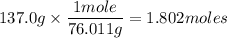
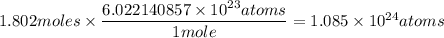

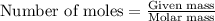
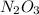
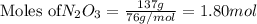
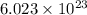 of particles
of particles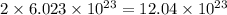 atoms of nitrogen
atoms of nitrogen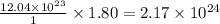 atoms of nitrogen.
atoms of nitrogen.



