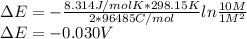(a) The change in cell voltage is 0.05V
(b) The change in cell voltage is 0.03V
Explanation:
Redox reaction : It is defined as the reaction in which the oxidation and reduction reaction takes place simultaneously.
Oxidation reaction : It is defined as the reaction in which a substance looses its electrons. In this, oxidation state of an element increases.
Reduction reaction : It is defined as the reaction in which a substance gains electrons. In this, oxidation state of an element decreases.
Further explanation:
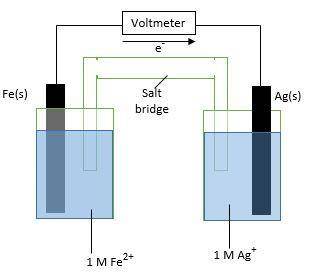
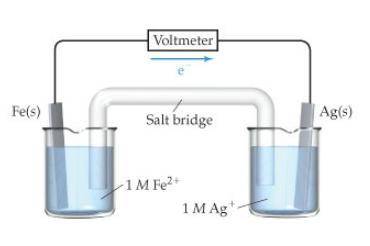
The image taken in context is attached below.
The standard reduction potentials for iron and silver are:
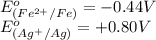
In the given cell, the oxidation occurs at an anode which is a negative electrode and the reduction occurs at the cathode which is a positive electrode.
From the standard reduction potentials we conclude that, the substance having highest positive 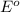 potential will always get reduced and will undergo reduction reaction.
potential will always get reduced and will undergo reduction reaction.
So, silver will undergo reduction reaction will get reduced. Iron will undergo oxidation reaction and will get oxidized.
The given cell reactions are:
Oxidation half reaction (anode): 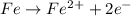
Reduction half reaction (cathode): 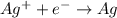
Thus, the anode and cathode will be 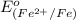
and 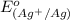 respectively.
respectively.
The overall cell reaction will be,
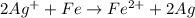
To calculate the 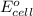 of the reaction, we use the equation:
of the reaction, we use the equation:
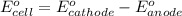
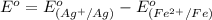
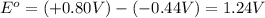
Now we have to calculate the cell potential.
Using Nernst equation :
![E_{cell}=E^o_{cell}-\frac{0.0592}{n}\log \frac{[Fe^{2+}]}{[Ag^{+}]^2}](/tpl/images/0487/5840/b460d.png)
where,
n = number of electrons in oxidation-reduction reaction = 2
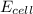 = emf of the cell = ?
= emf of the cell = ?
Now put all the given values in the above equation, we get:
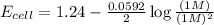

Thus, the emf of cell potential is 1.24 V
Part (a):
The ion concentrations in the cathode half-cell 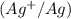 are increased by a factor of 10 from 1 M to 10 M.
are increased by a factor of 10 from 1 M to 10 M.
The emf of the cell potential will be,
Using Nernst equation :
![E_{cell}=E^o_{cell}-\frac{0.0592}{n}\log \frac{[Fe^{2+}]}{[Ag^{+}]^2}](/tpl/images/0487/5840/b460d.png)
where,
n = number of electrons in oxidation-reduction reaction = 2
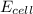 = emf of the cell = ?
= emf of the cell = ?
Now put all the given values in the above equation, we get:
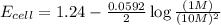

The change in cell voltage will be,
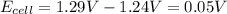
Thus, the change in cell voltage is 0.05V
Part (b):
The ion concentrations in the anode half-cell 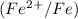 are increased by a factor of 10 from 1 M to 10 M.
are increased by a factor of 10 from 1 M to 10 M.
The emf of the cell potential will be,
Using Nernst equation :
![E_{cell}=E^o_{cell}-\frac{0.0592}{n}\log \frac{[Fe^{2+}]}{[Ag^{+}]^2}](/tpl/images/0487/5840/b460d.png)
where,
n = number of electrons in oxidation-reduction reaction = 2
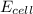 = emf of the cell = ?
= emf of the cell = ?
Now put all the given values in the above equation, we get:
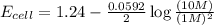

The change in cell voltage will be,
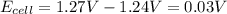
Thus, the change in cell voltage is 0.03V
Learn more:
Spontaneity of reaction; (answer by Kobenhavn)
Standard reduction potential; (answer by RomeliaThurston)
Keywords:
Nernst equation, standard reduction potential, spontaneity of the reaction.
















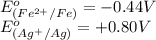
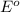 potential will always get reduced and will undergo reduction reaction.
potential will always get reduced and will undergo reduction reaction.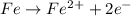
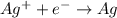
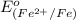
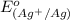 respectively.
respectively.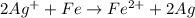
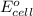 of the reaction, we use the equation:
of the reaction, we use the equation: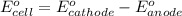
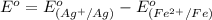
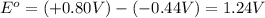
![E_{cell}=E^o_{cell}-\frac{0.0592}{n}\log \frac{[Fe^{2+}]}{[Ag^{+}]^2}](/tpl/images/0487/5840/b460d.png)
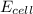 = emf of the cell = ?
= emf of the cell = ?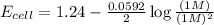

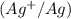 are increased by a factor of 10 from 1 M to 10 M.
are increased by a factor of 10 from 1 M to 10 M.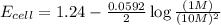

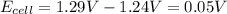
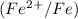 are increased by a factor of 10 from 1 M to 10 M.
are increased by a factor of 10 from 1 M to 10 M.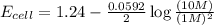

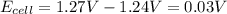

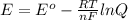
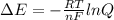
![Fe(s) - Fe^{2+}(aq)+2e [anode]\\2*(Ag^+(aq)+e - Ag(s)) [cathode]\\Fe(s)+2Ag^+(aq)-Fe^{2+}(aq)+2Ag(s) [overall]](/tpl/images/0487/5840/ad0ad.png)
![Q=\frac{[Fe^{2+}]}{[Ag^+]^2}](/tpl/images/0487/5840/f6f7d.png)
![\Delta E = - \frac{RT}{nF} ln \frac{[Fe^{2+}]}{[Ag^+]^2}](/tpl/images/0487/5840/41bf1.png)
![[Fe^{2+}]=10M](/tpl/images/0487/5840/2501c.png) and assume that T=298.15 K
and assume that T=298.15 K