
Chemistry, 29.01.2020 04:49, kimlyn58p0wyn0
If a given power plant released so2 gas with a volume v of 1200 m3 at a density ρ of 2.86 kg/m3 at standard pressure and temperature, how many moles n of so2 are released? the atomic weight of sulfur is 32.07 u and the atomic weight of oxygen is 16.00 u.

Answers: 2
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 09:00, miller5452
Acrystal that absorvd water from air is (blank)a. aqueousb. homogenousc. hygroscopicd. efflorescent
Answers: 1

Chemistry, 22.06.2019 10:30, cheyennecarrillo14
If you add 5.00 ml of 0.100 m sodium hydroxide to 50.0 ml of acetate buffer that is 0.100 m in both acetic acid and sodium acetate, what is the ph of the resulting solution? acetic acid: ka = 1.8. x 10-5
Answers: 1
Do you know the correct answer?
If a given power plant released so2 gas with a volume v of 1200 m3 at a density ρ of 2.86 kg/m3 at s...
Questions in other subjects:


English, 01.11.2020 14:00


Mathematics, 01.11.2020 14:00

Social Studies, 01.11.2020 14:00

Social Studies, 01.11.2020 14:00

Mathematics, 01.11.2020 14:00

Social Studies, 01.11.2020 14:00


Mathematics, 01.11.2020 14:00


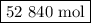
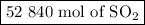 .
.




