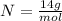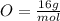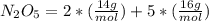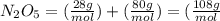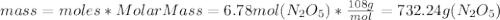Chemistry, 18.10.2019 10:20, ebonsell4910
What is the mass (in grams) of 6.78 moles of dinitrogen pentoxide

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:30, SchoolFirst9811
The scheme below is from a series of reactions that are part of a synthesis of vitamin a. answer the following questions with reference to this scheme. (i) what is "reagent a"? (ii) draw a step-by-step mechanism which explains the formation of compound c from compound b (iii) which reagents would you use to form compound e from compounds c and d (reagents b and c)? for each reagent suggested above in (ii) explain the role of the reagent in the reaction to (iv) form compound e. you may wish to do this by drawing a mechanism. 1. addition of reagent a но reagent a 2. н, о" thо oh нон-с compound a. compound b. compound c .ch-оh 1. reagent b "сно 2. reagent c сh oh compound e. compound d.
Answers: 2

Chemistry, 22.06.2019 13:30, justinerodriguz2878
What are the major types of a chemical compound
Answers: 2


Chemistry, 23.06.2019 01:30, sanchezvazquez0123
In which phase of mitosis do the spindle fibers pull the chromosomes apart to opposite sides of the cell ?
Answers: 1
Do you know the correct answer?
What is the mass (in grams) of 6.78 moles of dinitrogen pentoxide...
Questions in other subjects:





Mathematics, 15.07.2019 04:30


English, 15.07.2019 04:30



Mathematics, 15.07.2019 04:30


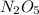 in 6.78 moles of the same compound.
in 6.78 moles of the same compound.