
Chemistry, 30.01.2020 10:44, dtrdtrdtrdtrdrt3415
Given the equation 2na+cl2 →2nacl how many moles of nacl will be produced from 59.0 grams of na, assuming cl is available in excess?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:20, whrjegt4jrnfdvj
1. suppose a reaction mixture, when diluted with water, afforded 300 ml of an aqueous solution of 30 g of the reaction product malononitrile [ch2(cn)2], which is to be isolated by extraction with ether. the solubility of malononitrile in ether at room temperature is 20.0 g/100 ml, and in water is 13.3 g/100 ml. what weight of malononitrile would be recovered by extraction with (a) three 100-ml portions of ether and (b) one 300-ml portion of ether? suggestion: for each extraction, let x equal the weight extracted into the ether layer. in part (a), the concentration in the ether layer is x/100 and in the water layer is (30 x)/300; the ratio of these quantities is equal to k 20/13.3.
Answers: 2

Chemistry, 22.06.2019 11:00, peternice2956
Ais a mountain created from eruptions of lava, ash, rocks, and hot gases.
Answers: 1

Chemistry, 22.06.2019 12:00, angtrevv
In a laboratory, 1.55mg of an organic compound containing carbon, hydrogen, and oxygen is burned for analysis. this combustion resulted in the formation of 1.45mg of carbon dioxide and .89 mg of water. what is the empirical formula for this compound?
Answers: 1

Chemistry, 22.06.2019 16:00, corcoranrobert1959
How do dying stars contribute to the formation of planets
Answers: 1
Do you know the correct answer?
Given the equation 2na+cl2 →2nacl how many moles of nacl will be produced from 59.0 grams of na, as...
Questions in other subjects:


English, 25.01.2022 02:50

Chemistry, 25.01.2022 02:50



Mathematics, 25.01.2022 02:50



Mathematics, 25.01.2022 02:50

English, 25.01.2022 02:50


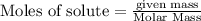
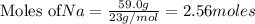
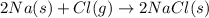
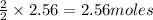 of NaCl
of NaCl




