

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, kingteron5870
Ionic compounds are made of ions, and yet the overall charge of an ionic compound is neutral. why?
Answers: 1



Do you know the correct answer?
Which of the following is true about the melting temperature of potassium chloride? the melting tem...
Questions in other subjects:

Physics, 10.10.2021 05:00

Mathematics, 10.10.2021 05:00

Biology, 10.10.2021 05:00





Spanish, 10.10.2021 05:00



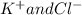 ions.
ions.




