Read the given equation. 2na + 2h2o → 2naoh + h2
during a laboratory experiment, a certain qu...

Chemistry, 03.11.2019 07:31, twalters88
Read the given equation. 2na + 2h2o → 2naoh + h2
during a laboratory experiment, a certain quantity of sodium metal reacted with water to produce sodium hydroxide and hydrogen gas. what was the initial quantity of sodium metal used if 6.30 liters of h2 gas were produced at stp?
a.) 10.3 grams
b.) 12.9 grams
c.) 14.7 grams
d.) 15.2 grams

Answers: 1
Other questions on the subject: Chemistry



Do you know the correct answer?
Questions in other subjects:

Mathematics, 07.07.2021 22:10

Mathematics, 07.07.2021 22:10






Chemistry, 07.07.2021 22:10

Mathematics, 07.07.2021 22:10

Mathematics, 07.07.2021 22:10


 .
.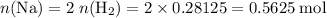 .
. .
.



