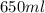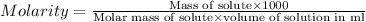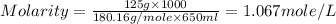Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, girly37
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 3


Do you know the correct answer?
A650.0 ml solution contains 125 grams of glucose (c6h12o6). if the molar mass of c6h12o6 is 180.16 g...
Questions in other subjects:


Chemistry, 31.03.2020 22:29

Biology, 31.03.2020 22:29

Mathematics, 31.03.2020 22:29




History, 31.03.2020 22:29


Social Studies, 31.03.2020 22:29