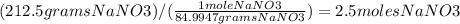Chemistry, 02.10.2019 08:50, itsjoke5550
Calculate the molarity of a solution in which 212.5g of nano3 are contained in 3.0 liters of solution.

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 13:50, hannahmyung1113
Amap that uses a range of colors and shading to represent the elevation, depth, or landscape of specific features on earth is a/an map.
Answers: 3

Chemistry, 22.06.2019 23:30, shukriabdisabrie
Match each statement with the state of matter it describes
Answers: 3

Chemistry, 23.06.2019 00:20, cmflores3245
4. propanol and isopropanol are isomers. this means that they have a) the same molecular formula but different chemical properties. b) different molecular formulas but the same chemical properties. c) the same molecular formula and the same chemical properties. d) the same molecular formula but represent different states of the compound
Answers: 3
Do you know the correct answer?
Calculate the molarity of a solution in which 212.5g of nano3 are contained in 3.0 liters of solutio...
Questions in other subjects:

Biology, 21.09.2019 01:00


History, 21.09.2019 01:00






English, 21.09.2019 01:00