
Answers: 1
Other questions on the subject: Chemistry


Chemistry, 23.06.2019 08:00, yddlex
Suppose a pair of chemical compounds a and b can react in two different ways: a + b -> c reaction 1 gives product c. a + b -> d reaction 2 gives product d. the following facts are known about the two reactions: . reaction 1 is endothermic and reaction 2 is exothermic. if a reaction vessel is charged (filled) with a and b , then at first d is produced faster than c. use these facts to sketch a qualitative reaction energy diagram for both reactions. note: because these sketches are only qualitative, the energies don? t have to be exact. they only have to have the right relationship to each other. for example, if one energy is less than another, that fact should be clear in your sketch.
Answers: 3

Chemistry, 23.06.2019 08:00, Robinlynn228
Pl what kind of reaction is this? nahco3 + h2o → co2 + naoh + h2o -composition -decomposition -single replacement -double replacement im leaning more toward single replacement. if im wrong can you explain whyy?
Answers: 1

Chemistry, 23.06.2019 10:30, GiuliAzevedo
The element chlorine has two stable isotopes, chlorine-35 with a mass of 34.97 amu and chlorine-37 with a mass of 36.95 amu. from the atomic weight of cl = 35.45 one can conclude that:
Answers: 2
Do you know the correct answer?
What is the mass of the hydrogen atoms in 1 mole of water...
Questions in other subjects:

Mathematics, 16.10.2020 16:01


Mathematics, 16.10.2020 16:01

English, 16.10.2020 16:01




Mathematics, 16.10.2020 16:01



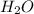
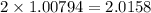 grams is the answer
grams is the answer




