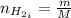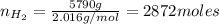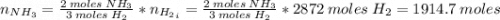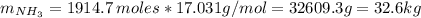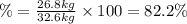Chemistry, 28.09.2019 15:10, victoriacarr638
Areaction vessel for synthesizing ammonia by reacting nitrogen and hydrogen is charged with 5.79 kg of h2 and excess n2. a total of 26.8 kg of nh3 are produced. what is the percent yield of the reaction?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:30, angemango3423
What is the empirical formula of c6h18o3? ch3o c2h5o c2h6o c2h5o5
Answers: 1

Chemistry, 22.06.2019 11:20, Jessicadiaz8602
Sodium nitrite (nano2) reacted with 2−iodooctane to give a mixture of two constitutionally isomeric compounds of molecular formula c8h17no2 in a combined yield of 88%. draw reasonable structures for these two isomers. click the "draw structure" button to launch the drawing utility. place the two compounds in the appropriate boxes below.
Answers: 1

Chemistry, 22.06.2019 12:30, quantamagic
Word equation for k(s)+h2o(l) yield koh(aq) + h2(g)
Answers: 1
Do you know the correct answer?
Areaction vessel for synthesizing ammonia by reacting nitrogen and hydrogen is charged with 5.79 kg...
Questions in other subjects:


Chemistry, 05.05.2020 03:01

History, 05.05.2020 03:01

Mathematics, 05.05.2020 03:01


Spanish, 05.05.2020 03:01


Mathematics, 05.05.2020 03:01

History, 05.05.2020 03:01


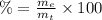 (2)
(2)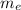 : is the experimental (or actual) mass = 26.8 kg
: is the experimental (or actual) mass = 26.8 kg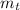 : is the theoretical mass
: is the theoretical mass