
Chemistry, 21.11.2019 18:31, ijustneedhelploll
On the basis of structure, which of the following solids has the highest melting point? question 4 options: sucrose ,table salt ,glucose , or butter

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:30, esnyderquintero
How do you lengthen a pattern piece? (family and consumer science, sewing)
Answers: 2



Chemistry, 23.06.2019 07:00, SMURFETTE86
Under what conditions will a gas be most likely to exhibit the ideal gas properties predicted by the ideal gas law? 1)high pressures and high temperature, because particles are forced closer together with higher kinetic energy, so intermolecular forces of attraction are weaker 2)high pressure and low temperature, because particles are forced closer together and moving slower, so the volume of the particles is less significant 3) low pressure and high temperature, because particles are spread farther apart and moving faster, so the intermolecular forces of attraction are weaker 4)low pressure and low temperature, because particles are spread farther apart with lower kinetic energy, so the volume of the particles is less significant
Answers: 2
Do you know the correct answer?
On the basis of structure, which of the following solids has the highest melting point? question 4...
Questions in other subjects:


Computers and Technology, 27.02.2021 22:10




Mathematics, 27.02.2021 22:10




Biology, 27.02.2021 22:10


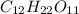 is a covalent compound which is formed by sharing of electrons and thus form a weak bond.
is a covalent compound which is formed by sharing of electrons and thus form a weak bond. 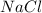 is a ionic compound formed by transfer of electrons and thus a strong bond is formed due to strong coloumbic forces.Thus large amount of energy is required to break the bonds and they have high melting points.
is a ionic compound formed by transfer of electrons and thus a strong bond is formed due to strong coloumbic forces.Thus large amount of energy is required to break the bonds and they have high melting points.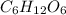 is a covalent compound which is formed by sharing of electrons and thus form a weak bond.
is a covalent compound which is formed by sharing of electrons and thus form a weak bond. 



