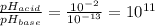Chemistry, 19.12.2019 11:31, erinloth123
Aph scale with ph values of some aqueous solutions. look at the figure showing the ph values of some familiar aqueous solutions. what is the difference between h+ concentration in an acidic solution such as lemon juice (ph 2) and a basic solution such as household bleach (ph 13)? a ph scale with ph values of some aqueous solutions. look at the figure showing the ph values of some familiar aqueous solutions. what is the difference between h+ concentration in an acidic solution such as lemon juice (ph 2) and a basic solution such as household bleach (ph 13)? the h+ concentration of lemon juice is higher than the h+ concentration of household bleach by a factor of 11. the h+ concentration of lemon juice is higher than the h+ concentration of household bleach by a factor of 1011 (100 billion). the h+ concentration of household bleach is higher than the h+ concentration of lemon juice by a factor of 1011 (100 billion).

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 23.06.2019 06:10, tammydbrooks43
How much would the freezing point of water decrease if 4 mol of nacl were added to 1 kg of water (kf=1.86 degrees c/(mol/kg) for water and i=2 for nacl a- 7.44 degrees c b- 14.88 c 3.72 d 1.86
Answers: 1

Chemistry, 23.06.2019 08:00, george27212
What is the temperature in kelvin of a gas if it is allowed to expand from 1.50 l to 4.50 l? the initial temperature is 10.0°c and pressure is constant throughout the change. which equation should you use? t2= v2/v1 t1 what is the final temperature? ⇒ 849 k these are the answers.
Answers: 1

Chemistry, 23.06.2019 16:30, mbonham481
Amodel of an atom is shown below. which element is represented by this model of an atom? boron, carbon, neon, or sodium?
Answers: 1
Do you know the correct answer?
Aph scale with ph values of some aqueous solutions. look at the figure showing the ph values of some...
Questions in other subjects:


Mathematics, 05.05.2020 14:54

Computers and Technology, 05.05.2020 14:54



History, 05.05.2020 14:54

Mathematics, 05.05.2020 14:54

Mathematics, 05.05.2020 14:54



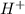 concentration of lemon juice is higher than the
concentration of lemon juice is higher than the 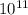 (100 billion)
(100 billion)![pH=-log[H^+]](/tpl/images/0425/9876/15713.png)
![2=-log[H^+]](/tpl/images/0425/9876/a01ca.png)
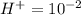
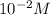 of
of ![13=-log[H^+]](/tpl/images/0425/9876/f4c07.png)
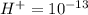
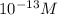 of
of