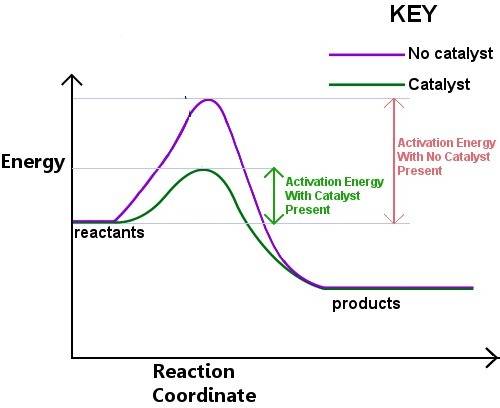
Chemistry, 22.06.2019 19:00, innocentman69
How does a catalyst increase the speed of a reaction? a. the catalyst eliminates the activated complex stage, allowing products to form immediately. b. the catalyst lowers the energy level of the reactants, making it easier for them to react. c. the catalyst makes it easier for the activated complex to form, lowering the activation energy. d. the catalyst raises the energy level of the products, making the reaction finish sooner. reset next

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:30, tntaylor862
Using data from seismic waves, geologists have learned that earth’s interior is made up of several
Answers: 1



Chemistry, 23.06.2019 01:30, yarrito20011307
Which conclusion fits the data in the table? a. heat chemically changes chocolate and margarine. b. all solids become liquid at 100°f. c. removing heat from a substance it to melt. d. matter may change shape when it is heated.
Answers: 1
Do you know the correct answer?
How does a catalyst increase the speed of a reaction? a. the catalyst eliminates the activated comp...
Questions in other subjects:



Mathematics, 07.06.2021 20:50

Mathematics, 07.06.2021 20:50

Mathematics, 07.06.2021 20:50



English, 07.06.2021 20:50

Mathematics, 07.06.2021 20:50

Mathematics, 07.06.2021 20:50