
Chemistry, 23.06.2019 03:30, HalpMahOnMahH0meW0rk
Calculate the ph of a .10m nh4cl solution. the kb value for nh3 is 1.8×10^-5

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 09:50, bridgetosanders
What are four significant sources of ghgs that come from wostem washington?
Answers: 2


Chemistry, 22.06.2019 21:20, jordan2875
If a simple machine aduces the strength of a force, what must be increased? the speed of the input force the work the simple machine performs the size of the simple machine the distance over which the force is applied
Answers: 1
Do you know the correct answer?
Calculate the ph of a .10m nh4cl solution. the kb value for nh3 is 1.8×10^-5...
Questions in other subjects:









Business, 11.09.2019 23:30

Computers and Technology, 11.09.2019 23:30


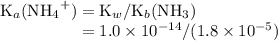
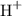 with the unit M (the same as
with the unit M (the same as 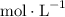 ).
).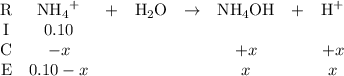
![\text{K}_a = [\text{NH}_4\text{OH}] \cdot [\text{H}^{+}] / [{\text{NH}_4}^{+}] \\\phantom{\text{K}_a} = x^{2} / (0.10 - x)](/tpl/images/0006/4800/9d0ac.png)
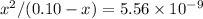
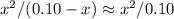
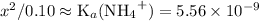
 .
.![[\text{H}^{+}] = x = 2.36 \times 10^{-5} \; \text{mol}\cdot \text{L}^{-1}](/tpl/images/0006/4800/5c4f5.png) ,
,![\text{pH} = -\log_{10}{[\text{H}^{+}] = 4.63](/tpl/images/0006/4800/bfdbc.png) .
.



