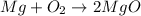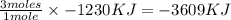Chemistry, 26.06.2019 15:00, trinity7265
Magnesium oxidizes via the reaction: 2 mg + o2 → 2 mgo the reaction has a △hrxn = -1203 kj. how much heat (in kj) is released when you completely react 3.000 moles of o2?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 21:30, MJyoungboy
Harry lives in a city, and he has a lung condition known as asthma. on certain days, harry has to stay inside because pollutants in the air make it difficult for him to breathe. which of these pollution sources are nonpoint sources that might bother harry if he goes outside? choose the two that apply.
Answers: 3

Chemistry, 23.06.2019 05:30, brianrodriguez2005
What is the body’s main processing system? it uses input from various parts to control voluntary and involutiontary movement. it’s composed of two main parts-the brain and spinal cord. a. nbs b. cns c. ans d. pns
Answers: 1

Do you know the correct answer?
Magnesium oxidizes via the reaction: 2 mg + o2 → 2 mgo the reaction has a △hrxn = -1203 kj. how muc...
Questions in other subjects:


English, 27.07.2019 03:00

History, 27.07.2019 03:00

English, 27.07.2019 03:00



Mathematics, 27.07.2019 03:00

Mathematics, 27.07.2019 03:00



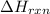 = -1230 KJ
= -1230 KJ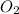 = 3 moles
= 3 moles