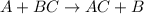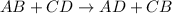Chemistry, 26.06.2019 20:30, johndacres8280
Which statement is correct about the reaction that occurs between calcium chloride and sodium carbonate? it is a single replacement reaction because two new compounds of calcium and sodium are formed. it is a double replacement reaction because calcium chloride and sodium carbonate exchange their non-metallic parts. it is a single replacement reaction because calcium replaces sodium within a compound as sodium is higher than calcium in the activity series. it is a double replacement reaction because the ions of calcium and sodium exchange places in an aqueous solution as calcium is higher than sodium in the activity series.

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 23.06.2019 02:20, theactualslash
In a chemical reaction, the final amount of the products is determined by the a. universal gas law b. law of definite proportions c. air pressure d. temperature e. none of the above me
Answers: 2

Chemistry, 23.06.2019 06:30, lwattsstudent
When microscope slides are stained to show blood cells, the small red blood cells that appear on the slides are much numerous than the large white blood cells. this supports the concept that
Answers: 1

Chemistry, 23.06.2019 08:30, vett072804
Benzonitrile (c6h5cn) is reduced to two different products depending on the reducing agent used. treatment with lithium aluminum hydride followed by water forms k, which has a molecular ion in its mass spectrum at 107 and the following ir absorptions: 3373, 3290, 3062, 2920, and 1600 cm-1. treatment with a milder reducing agent forms l, which has a molecular ion in its mass spectrum at 106 and the following ir absorptions: 3086, 2850, 2820, 2736, 1703, and 1600 cm-1. l shows fragments in its mass spectrum at m/z = 105 and 77. propose structures for k and l and choose an explanation for how this could be concluded.
Answers: 3
Do you know the correct answer?
Which statement is correct about the reaction that occurs between calcium chloride and sodium carbon...
Questions in other subjects:

Mathematics, 28.02.2020 01:25