Chemistry, 27.06.2019 08:30, daryondaniels28
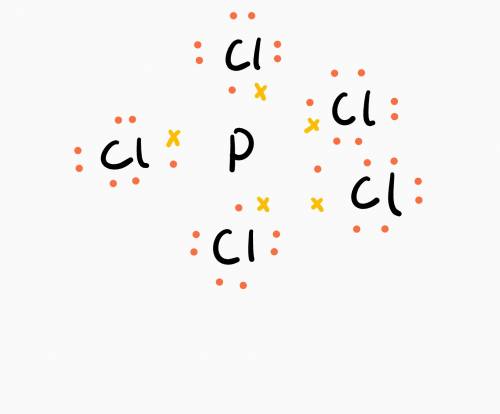
Many elements in the third row and beyond in the periodic table may form more than four bonds and thus appear to have "expanded octets." phosphorus and sulfur, for example, may form five and six covalent bonds. count up the total number of valence electrons in pcl5 and draw its lewis structure. how many valence electrons are "counted" toward the central p atom?

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 19:20, halledoll2002
Anyone who's in connections academy chemistry b have the factors that affect the rate of a reaction portfolio already done?
Answers: 3
Do you know the correct answer?
Many elements in the third row and beyond in the periodic table may form more than four bonds and th...
Questions in other subjects:


English, 28.10.2020 21:30

Mathematics, 28.10.2020 21:30

History, 28.10.2020 21:30


English, 28.10.2020 21:30


Chemistry, 28.10.2020 21:40