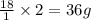Chemistry, 28.06.2019 23:00, mercydiaz84
The following chemical equation describes the chemical reaction of hydrogen gas and oxygen gas to create water 2h2 + o2 = 2h2o use what you know about molar relationships to explain how scientists can predict the amount of water produced if they know the amounts of hydrogen and oxygen gases they have to react.

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 06:30, yolo123321
The following reaction shows sodium carbonate reacting with calcium hydroxide. na2co3 + ca(oh)2 → naoh + caco3 how many grams of naoh are produced from 20.0 grams of na2co3? (molar mass of na = 22.989 g/mol, c = 12.01 g/mol, o = 15.999 g/mol, ca = 40.078 g/mol, h = 1.008 g/mol) 12.2 grams 15.1 grams 24.4 grams 30.2 grams
Answers: 2

Chemistry, 22.06.2019 11:00, RidhaH
Which statement correctly identifies the scientific question and describes why the question is scientific? question 1 refers to the supernatural. question 2 reflects a moral or social value. question 3 refers to something that can be measured. question 4 reflects a question that can’t be observed.
Answers: 1
Do you know the correct answer?
The following chemical equation describes the chemical reaction of hydrogen gas and oxygen gas to cr...
Questions in other subjects:



Mathematics, 08.03.2020 23:45



Mathematics, 08.03.2020 23:45


Mathematics, 08.03.2020 23:45



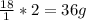 .
.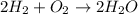
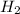 weighs 2 g.
weighs 2 g.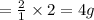
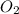 weighs 32 g
weighs 32 g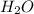 weighs = 18g
weighs = 18g