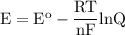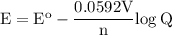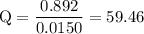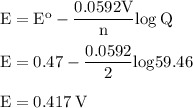Chemistry, 30.06.2019 19:30, joannegrace4898
Calculate the cell potential for the following reaction as written at 25.00 °c, given that [cr2 ] = 0.892 m and [fe2 ] = 0.0150 m. standard reduction potentials can be found here. cr(s)+fe2+(aq)< > cr2+(aq)+fe(s)

Answers: 1
Similar questions

Chemistry, 28.06.2019 03:40, cristabean87
Answers: 2

Chemistry, 30.06.2019 11:30, trvptierra
Answers: 1

Chemistry, 17.07.2019 20:30, thasen31
Answers: 1
Do you know the correct answer?
Calculate the cell potential for the following reaction as written at 25.00 °c, given that [cr2 ] =...
Questions in other subjects:

Chemistry, 20.10.2020 01:01




Biology, 20.10.2020 01:01