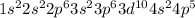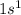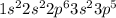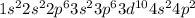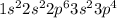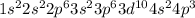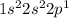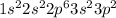Explanation:
Covalent bonds are defined as the bonds which are formed by the sharing of electrons between the atoms. They are usually formed by non-metals.
The number of covalent bonds are predicted by the number of valence electrons.
Elements belonging to Period 3 and above will form covalent bonds same as the number of valence electron because of the presence of d-orbitals.
Elements belonging to period 2 will form covalent bonds = (8 - number of valence electrons)
For the given elements:
Bromine is the 35th element which belong to period 4. The electronic configuration of 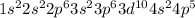
This element has 7 valence electrons and will form 7 covalent bonds.
Hydrogen is the 1st element which belong to period 1. The electronic configuration of 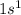
This element has 1 valence electrons and will form 1 covalent bond.
Chlorine is the 17th element which belong to period 3. The electronic configuration of 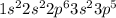
This element has 7 valence electrons and will form 7 covalent bonds.
Germanium is the 32nd element which belong to period 4. The electronic configuration of 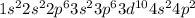
This element has 4 valence electrons and will form 4 covalent bonds.
Sulfur is the 16th element which belong to period 3. The electronic configuration of 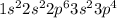
This element has 6 valence electrons and will form 6 covalent bonds.
Arsenic is the 33rd element which belong to period 4. The electronic configuration of 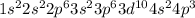
This element has 5 valence electrons and will form 5 covalent bonds.
Boron is the 5th element which belong to period 2. The electronic configuration of 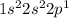
This element has 3 valence electrons. It will form (8 - 3) = 5 covalent bonds.
Silicon is the 14th element which belong to period 3. The electronic configuration of 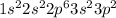
This element has 4 valence electrons and will form 4 covalent bonds.