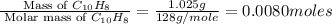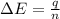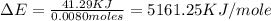Chemistry, 01.07.2019 03:00, Jadamachado45
When 1.025 g of naphthalene (c10h8) burns in a bomb calorimeter, the temperature rises from 24.25°c to 32.33°c. find δerxn for the combustion of naphthalene. the heat capacity of the calorimeter is 5.11 kj/°c?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:10, CauseWhyNot6235
What can be added to the examples section of each circle? endothermic: ice melting into water, and a heat pack becoming warm exothermic: a glow stick glowing, and fireworks exploding endothermic: ice melting into water, and an instant ice pack turning cold exothermic: fireworks exploding, and gasoline burning endothermic: a glow stick glowing, and a heat pack becoming warm exothermic: an instant ice pack turning cold, and ice melting into water endothermic: gasoline burning, and an instant ice pack turning cold exothermic: ice melting into water, and an instant ice pack turning cold
Answers: 1

Chemistry, 22.06.2019 20:30, huangjianhe135
The activation energy for the reaction no2(g)+co2(g)⟶no(g)+co(g) is ea = 300 kj/mol and the change in enthalpy for the reaction is δh = -100 kj/mol . what is the activation energy for the reverse reaction?
Answers: 3

Do you know the correct answer?
When 1.025 g of naphthalene (c10h8) burns in a bomb calorimeter, the temperature rises from 24.25°c...
Questions in other subjects:


Mathematics, 23.04.2021 01:00



Mathematics, 23.04.2021 01:00

Chemistry, 23.04.2021 01:00


Physics, 23.04.2021 01:00


Mathematics, 23.04.2021 01:00


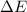 for the combustion of naphthalene is
for the combustion of naphthalene is 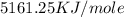
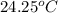
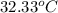
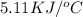
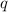
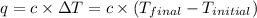
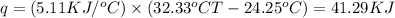
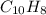 =
=