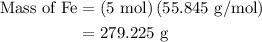Chemistry, 01.07.2019 13:00, amycressey1970
The following data was collected when a reaction was performed experimentally in the laboratory. fe2o3 al al2o3 fe starting amount in reaction 3 moles 5 moles ? ? determine the maximum amount of fe that was produced during the experiment. explain how you determined this amount.

Answers: 1
Other questions on the subject: Chemistry



Do you know the correct answer?
The following data was collected when a reaction was performed experimentally in the laboratory. fe2...
Questions in other subjects:




Chemistry, 24.09.2019 19:00




Health, 24.09.2019 19:00

Social Studies, 24.09.2019 19:00

Mathematics, 24.09.2019 19:00


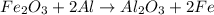
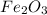 = 3 moles
= 3 moles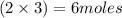 of Al
of Al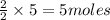 of Fe.
of Fe.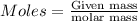
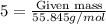
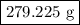 of Fe is produced during the given experiment.
of Fe is produced during the given experiment.
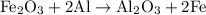
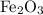 reacts with two moles of Al to produce one mole of
reacts with two moles of Al to produce one mole of 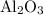 and two moles of Fe.
and two moles of Fe.
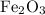 and 5 moles of Al. Therefore the amount of Fe produced by 3 moles of
and 5 moles of Al. Therefore the amount of Fe produced by 3 moles of 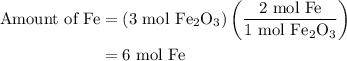
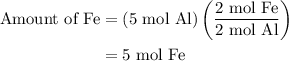
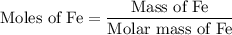 ...... (1)
...... (1) 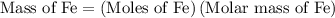 ...... (2)
...... (2)