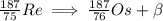Chemistry, 23.09.2019 13:10, golderhadashaowtatz
Chemistry! ! !
1. which of the following solutions would have a ph value greater than 7?
a. [oh-] = 2.4 × 10^-2 m
b. [h3o+] =1.53 × 10^-2 m
c. 0.0001 m hcl
d. [oh^-] = 4.4 × 10^-9 m
2. consider the following equation for an equilibrium system: (attached)
which concentration(s) would be included in the denominator of the equilibrium constant expression?
a. pb(s), co2(g), and so2(g)
b. pbs(s), o2(g), and c(s)
c. o2(g), pb(s), co2(g), and so2(g)
d. o2(g)
3. the oxidation number of the sulfur atom in the so^2- 4 ion is
a. +2.
b. -2.
c. +6.
d. +4.
4. in the following reaction, which is the oxidizing agent? (attached - reaction starts with agno2)
a. agno2
b. cl2
c. koh
d. kcl
5. complete the following nuclear equation (attached w/ choices)
6. which two particles have the same mass but opposite charge?
a. a beta particle and a positron
b. a neutron and a proton
c. a proton and an electron
d. an alpha particle and a proton
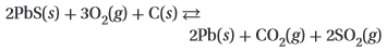
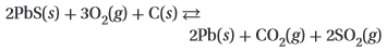

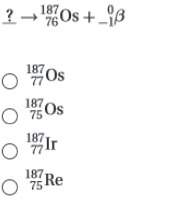

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 13:30, suemmimonjaras8374
The atomic number, or number, is the described as the number of in the nucleus of an chemical element.
Answers: 1

Chemistry, 22.06.2019 13:30, citlalli30
1) which of the following is the best example of a physical change? a) sugar dissolving in tea b) firefly glowing 2) in the combustion of ethane, what is/are the reactants? c2h6 + o2 ==> co2 + h2o a) c2h6 and o2 b) co2 and c2h6
Answers: 2
Do you know the correct answer?
Chemistry! ! !
1. which of the following solutions would have a ph value greater than 7? <...
1. which of the following solutions would have a ph value greater than 7? <...
Questions in other subjects:







Mathematics, 07.10.2019 08:00



Social Studies, 07.10.2019 08:00


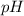 and
and 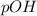 is given by
is given by 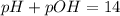 , We can use
, We can use ![pH=-log[H^+]](/tpl/images/0254/9117/15713.png) and
and ![pOH= -log[OH^-]](/tpl/images/0254/9117/291d4.png) . In a we are given the concentration of [OH] and so we use that to find the pOH, then from pOH we can find the pH.
. In a we are given the concentration of [OH] and so we use that to find the pOH, then from pOH we can find the pH.![pOH= -log [2.4 \times 10^-^2] = 1.62\\\\pH = 14 - 1.62= 12.38](/tpl/images/0254/9117/a7174.png)
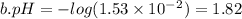
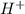 and 0.0001
and 0.0001 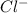 , hence
, hence ![pH= -log[0.0001] = 4](/tpl/images/0254/9117/358f1.png)
![d.pOH= -log [4.4 \times 10^-9] = 8.36\\\\pH= 14- 8.36 = 5.64](/tpl/images/0254/9117/bb698.png)
![a.[OH^-] = 2.4 \times 10^-^2](/tpl/images/0254/9117/6eb94.png) has a pH 12.36 which is greater than 7
has a pH 12.36 which is greater than 7 .
. ![K= \frac{[products]}{[reactants]}](/tpl/images/0254/9117/375cf.png) .
.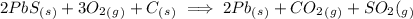
![K= \frac{[CO_2][SO_2]}{[O_2]}](/tpl/images/0254/9117/6da50.png)
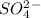 is -2 and in all its compounds oxygen has -2 charge, hence
is -2 and in all its compounds oxygen has -2 charge, hence
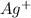 and
and  are spectactor ions in this reaction.
are spectactor ions in this reaction.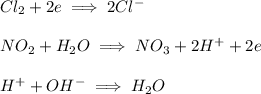
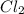 gains electrons to become
gains electrons to become