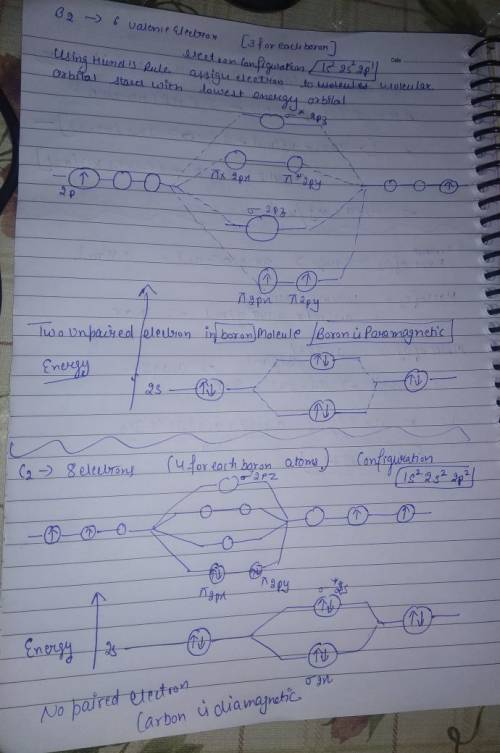
Chemistry, 02.07.2019 17:30, ramentome7542
Part a by drawing molecular orbital diagrams for b2, c2, n2, o2, and f2, predict which of these homonuclear diatomic molecules are magnetic.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:30, officialrogerfp3gf2s
How to solve 4 nh3(g) + 5 o2(g) > 4 no(g) + 6 h2o(g) in chemistry
Answers: 1

Chemistry, 21.06.2019 18:20, datboyjulio21
Complete the table for ion charge based upon their losing or gaining electrons in the outer shell. (use the periodic table as necessary.) group most likely ionic charge # of valence electrons i +1 ii +2 iii +3 iv +4 or -4 v -3 vi -2 vii -1 viii 0
Answers: 2

Chemistry, 22.06.2019 08:00, wizz4865
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

Chemistry, 22.06.2019 15:30, ricardotavarez6
How does a large body of water, such as the ocean, influence climate?
Answers: 1
Do you know the correct answer?
Part a by drawing molecular orbital diagrams for b2, c2, n2, o2, and f2, predict which of these homo...
Questions in other subjects:

Mathematics, 08.07.2019 16:00


Mathematics, 08.07.2019 16:00

Mathematics, 08.07.2019 16:00

Mathematics, 08.07.2019 16:00

History, 08.07.2019 16:00


History, 08.07.2019 16:00