Answers:
1. 3-ethyl-3-methylheptane; 2. 2,2,3,3-tetramethylpentane; 3. hexa-2,4-diene.
Explanation:
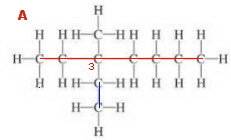
Structure 1
Identify and name the longest continuous chain of carbon atoms (the main chain has 7 C; ∴ base name = heptane).
Identify and name all the substituents [a 1C substituent (methyl) and a 2C substituent (methyl).
Number the main chain from the end closest to a substituent.
Identify the substituents by the number of the C atom on the main chain. Use hyphens between letters and numbers (3-methyl, 3-ethyl).
Put the names of the substituents in alphabetical order in front of the base name with no spaces (3-ethyl-3-methylheptane)
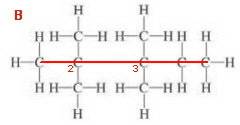
Structure 2
5C. Base name = pentane
Four methyl groups.
Number from the left-hand end.
If there is more than one substituent of the same type, identify each substituent by its locating number and use a multiplying prefix to show the number of each substituent. Use commas between numbers (2,2,3,3-tetramethyl).
The name is 2,2,3,3-tetramethylpentane.
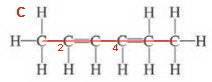
Structure 3
Identify and name the longest continuous chain of carbon atoms that passes through as many double bonds as possible. Drop the -ne ending of the alkane to get the root name hexa-.
(No substituents).
Number the main chain from the end closest to a double bond.
If there is more than one double bond use a multiplying prefix to indicate the number of double bonds (two double bonds = diene) and use the smaller of the two numbers of the C=C atoms as the double bond locators (2,4-diene)
Put the functional group name at the end of the root name (hexa-2,4-diene).
Note: The name 2,4-hexadiene is acceptable, but the Preferred IUPAC Name puts the locating numbers as close as possible in front of the groups they locate.