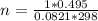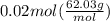Chemistry, 03.07.2019 14:30, corbinfisher
If 495 milliliters of carbon dioxide at 25°c and 101.3 kilopascals reacts with excess water, what is the theoretical yield of carbonic acid?

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 23.06.2019 10:20, Thejollyhellhound20
Based on the equation, how many grams of br2 are required to react completely with 29.2 grams of alcl3? alcl3 + br2 → albr3 + cl2 48.7 grams 52.6 grams 56.7 grams 61.3 grams
Answers: 3

Chemistry, 23.06.2019 10:40, 1r32tgy5hk7
Question 17 hydrogen is manufactured on an industrial scale by this sequence of reactions: +ch4gh2og ⇌ +cog3h2g k1 +cogh2og ⇌ +co2gh2g k2 the net reaction is: +ch4g2h2og ⇌ +co2g4h2g k write an equation that gives the overall equilibrium constant k in terms of the equilibrium constants k1 and k2. if you need to include any physical constants, be sure you use their standard symbols, which you'll find in the aleks calculator.
Answers: 2
Do you know the correct answer?
If 495 milliliters of carbon dioxide at 25°c and 101.3 kilopascals reacts with excess water, what is...
Questions in other subjects:


Mathematics, 05.05.2020 19:33





Mathematics, 05.05.2020 19:33

Mathematics, 05.05.2020 19:33



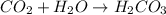
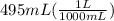 = 0.495 L
= 0.495 L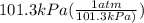
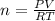
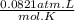 .
.