Chemistry, 04.07.2019 04:30, Homepage10
32) in the synthesis reaction of magnesium and chlorine, two chlorine atoms react with one magnesium atom to form the ionic compound, mgcl2. what is the best explanation for this reaction?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:00, Homepage10
Theoretically, which metal should be the most reactive? hydrogen lithium francium fluorine
Answers: 1

Chemistry, 21.06.2019 22:30, mimireds5419
1. combine iron and copper (ii) sulfate solution. (hint: iron will form the iron (iii) ion) fe + cuso4 → 2. combine lead (ii) nitrate and potassium iodide solutions. pb(no3)2+ kl → 3. combine magnesium metal and hydrochloric acid solution. mg + hcl → 4. electrolysis (splitting) of water. h2o → 5. burning magnesium. mg + o2 →
Answers: 3

Chemistry, 22.06.2019 04:30, salvadorperez26
Suppose that during that icy hot lab 65,000 j of energy were transferred to 450 g of water at 20°c what would have have been the final temperature of the water
Answers: 2
Do you know the correct answer?
32) in the synthesis reaction of magnesium and chlorine, two chlorine atoms react with one magnesium...
Questions in other subjects:


Mathematics, 26.06.2019 04:20

Social Studies, 26.06.2019 04:20






Chemistry, 26.06.2019 04:20

Mathematics, 26.06.2019 04:20


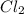 , The balanced equation for the synthesis reaction of magnesium and chlorine to form magnesium chloride is written as:
, The balanced equation for the synthesis reaction of magnesium and chlorine to form magnesium chloride is written as: